
The light curve of the supernova is associated with the decay of nickel-56 to cobalt-56 and then to iron-56. Difference spectrum between unmodified Ni and TA modified Ni, at pH 5 (1), nickel tartrate (2). The effects of temperature on the structure of ceramics were. XPS spectra in the C1s (a) and O1s (b) regions. One of its isotopes, nickel-56, is produced in type II supernova. Bulk Y2NiMnO6 samples were prepared by thermal decomposition technique at 800 C for 6 hours. corresponding to the metallic alloy (Ni, Cr, Fe) are not detected (see for example Fig. It also makes up crucibles that are used in chemical laboratories. The XPS spectra of the oxidised samples (before sputtering). Anchoring of a nickel(II) Schiff base complex onto activated carbon mediated by cyanuric chloride. Nickel is also used in the five-cent coins in the United States and Canada (called nickels). A frequent area of analysis failure involves fitting XPS spectra without consideration of physics and chemistry of the spectrum. Nickel steel is used for armor plates and vaults. Most of the nickel consumed in the Western World is used to make austenitic stainless steel. The majority of the supply of nickel is believed to be located in the Earth’s core, while Canada, Russia, New Caledonia, Australia, Cuba, and Indonesia house accessible deposits of nickel.
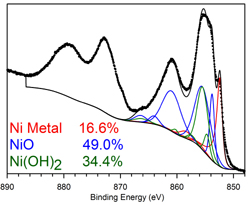
The black nickel survey scan exhibits the main core-level peaks for O 1s, C 1s, N 1s, S 2p, Zn 2p, and Ni 2p, centered at binding energies 531.6, 284.5, 397.9-400, 162.7, 1021.6, and 852.9-855.7 eV, respectively. The Fe 2p, Cr 2p, Ni 2p and Mo 3d XPS signals recorded after oxidation at. The C1s (284.6 eV) peak was used to adjust the position of the XPS spectra. The consumption of nickel can be traced back to 3500 BC. In SPS alone, orthopedic stainless steel shows a small anodic peak at -0.4.
